Technical Handbook for Marine Biodiesel |
||
|
Technical Handbook for Marine Biodiesel In Recreational Boats by Randall von Wedel, Ph.D. CytoCulture International, Inc. Point Richmond, CA
Second Edition April 22, 1999 Marine Biodiesel and Education Project for San Francisco Bay and Northern California Prepared for the National Renewable Energy Laboratory U.S. Department of Energy Subcontract No. ACG-7-16688-01 under Prime Contract No. DE-AC36-83CH10093 Contents Emissions Reductions with Biodiesel Lower Impact on Marine Environment Engine Performance: Mechanical Advantages Safety and Aesthetic Advantages of Biodiesel
Appendices and Schematic of Transesterification Process
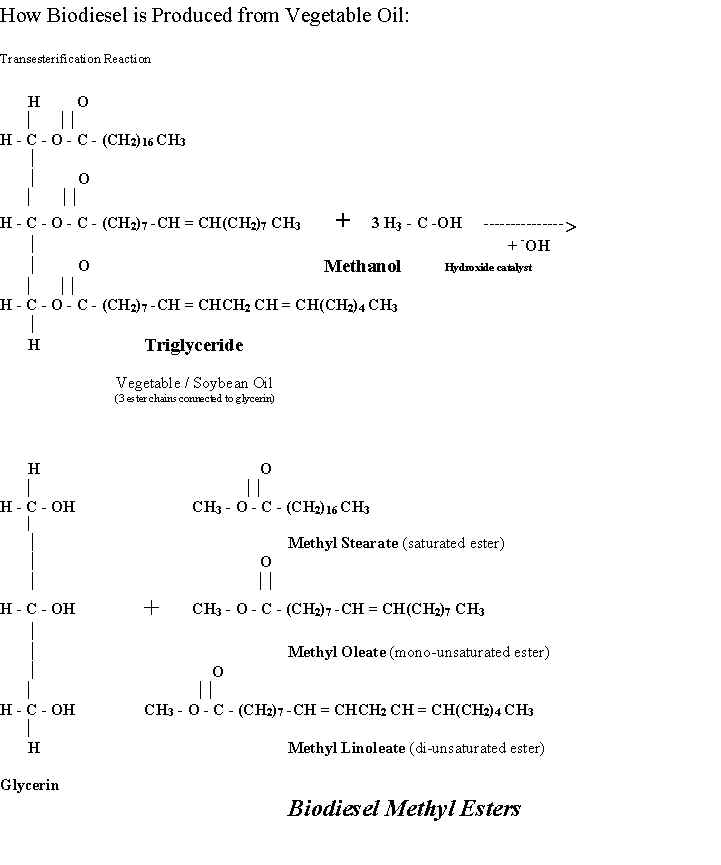
Technical Handbook for Marine Biodiesel This handbook has been prepared to provide practical information on Biodiesel to owners of recreational boats powered with diesel engines. The report summarizes research work and field observations collected over the past five years from the U.S. and Europe. The handbook is intended to be relatively comprehensive without being overly detailed. References are cited to guide the reader in pursuing specific topics in more depth. The appendices contain support documentation and articles on marine Biodiesel. BIODIESEL: Fuel Additive made from Vegetable Oil Biodiesel is a clean-burning diesel fuel additive produced from soybean and other vegetable oils instead of petroleum. Biodiesel is marketed in California for use in marine compression ignition (diesel) engines to enhance engine combustion performance, improve engine lubrication, and reduce air and water pollution caused by the exhaust. Biodiesel blends operate in diesel engines, from light to heavy-duty, just like petroleum diesel fuel. No engine conversions are required at all, unless an engine has old fuel lines. Biodiesel and a 20% blend of Biodiesel in petroleum diesel are DOE-designated alternative fuels. Biodiesel is registered as a fuel additive with the Environmental Protection Agency (EPA). Biodiesel and the 20% blend meet clean diesel standards established by the California Air Resources Board (CARB), particularly since the Biodiesel contains no sulfur and no aromatics. The National Biodiesel Board maintains specifications for Biodiesel and has worked with the American Society for Testing and Materials (ASTM) to develop a provisional ASTM standard for Biodiesel production in the U.S.. In 1998, Biodiesel as a 20% blend ("B-20") with petroleum diesel was designated an "alternative fuel" under the Energy Policy Act. This designation allows government fleet services to purchase the B-20 blend for operation in normal diesel vehicles and receive credit for those vehicles equivalent to other DOE-approved multi-fuel vehicles. As a result, Biodiesel can now compete with other alternative fuels and clean-air options for urban transit fleets and government vehicles across the country. For the marine market, this DOE designation should encourage more Biodiesel production and, eventually, lower prices for consumers. Biodiesel is Produced from Vegetable Oils by a Process called Transesterification (see Appendix for schematic of process) Biodiesel is produced from vegetable oils by converting the triglyceride oils to methyl (or ethyl) esters with a process known as transesterification. The transesterification process reacts alcohol with the oil to release three "ester chains" from the glycerin backbone of each triglyceride. The reaction requires heat and an strong base catalyst (e.g., hydroxide or lye), to achieve complete conversion of the vegetable oil into the separated esters and glycerin. The glycerin can be further purified for sale to the pharmaceutical and cosmetic industries. The mono-alkyl esters become the Biodiesel, with one-eighth the viscosity of the original vegetable oil. Each ester chain, usually 18 carbons in length for soy esters, retains two oxygen atoms forming the "ester" and giving the product its unique combustion qualities as an oxygenated vegetable based fuel. Biodiesel is nearly 10% oxygen by weight. Petroleum diesel, in contrast, is made up of hundreds of different hydrocarbon chains (roughly in the range of 14-18 carbons in length), with residues of sulfur and crude oil remaining. Diesel fuel sold today, even "low sulfur, low aromatic" diesel, contains 20-24% aromatics (benzene, toluene, xylenes, etc.) which are toxic, volatile compounds responsible for the fire/health hazards and pollution associated with petroleum diesel. Sailboats with Auxiliary Diesel Engines Recreational sailboats powered by auxiliary diesel engines have proven to be a reliable and high profile market for Biodiesel. In 1997, CytoCulture surveyed 100 recreational boaters in the San Francisco Bay area and found that 97% of the vessels using Biodiesel from 1993 to 1997 were sailboats. Most of the boats were in the 30 to 50 foot range, and most had smaller diesel engines (12-50 HP) that consumed relatively little fuel. Sailboaters tend to be more conscious of environmental concerns, they are sensitive to smoke and odor from engine exhaust, and they are more inclined than power boaters to pay for premium diesel fuel since they typically consume only 10-50 gallons a year. Future marine markets in which the benefits of using Biodiesel would outweigh the costs include charter boats, water taxis, dive boats, small ferries, government boats and research vessels. There should be particular emphasis on using Biodiesel in boats operating on lakes, rivers and confined bays that are more sensitive to air and water pollution. Aside from sailing with the wind, vegetable oil Biodiesel is the easiest, cleanest and most efficient transformation of solar energy to produce kinetic energy for mechanical power in boats. Recommended Blending Ratios for Biodiesel in Boat Fuel Biodiesel mixes easily with diesel as a fuel additive for use in blends of up to 20% with regular petroleum diesel. Add 5 gallons (one 5-gallon container) of Biodiesel to every 20 gallons of petrodiesel to achieve a 20% blend, or use the blending chart printed on the container back label. Biodiesel mixes quickly with petrodiesel once the boat is moving. Biodiesel is a little heavier than the petroleum with has a specific gravity of 0.87 compared to 0.79-0.80 typical of reformulated petrodiesels. Higher concentrations, up to 100% (neat) Biodiesel, are used in Europe to operate diesel engines in boats and vehicles with good performance results and excellent emissions reductions. However, until new Federal and State laws defining diesel fuel specifications are mandated to accommodate the unique properties of vegetable methyl esters, Biodiesel will only be sold as an additive for use in boat engines at ratios not to exceed 20%. In France, all diesel sold for vehicle fuel in the entire country ranges from 1% up to 5% rapeseed Biodiesel in a blend and some urban buses routinely operate on a 30% blend. In Germany, where the price of Biodiesel (tax exempt) is similar to petroleum diesel (with taxes), over 350 fuel stations offer Biodiesel for sale to motorists and Biodiesel is used in tour boats on their lakes. EMISSIONS REDUCTIONS WITH BIODIESEL Since Biodiesel is made entirely from vegetable oil, it does not contain any sulfur, aromatic hydrocarbons, metals or crude oil residues. The absence of sulfur means a reduction in the formation of acid rain by sulfate emissions which generate sulfuric acid in our atmosphere. The reduced sulfur in the blend will also decrease the levels of corrosive sulfuric acid accumulating in the engine crankcase oil over time. The lack of toxic and carcinogenic aromatics (benzene, toluene and xylene) in Biodiesel means the fuel mixture combustion gases will have reduced impact on human health and the environment. The high cetane rating of Biodiesel (ranges from 49 to 62) is another measure of the additive's ability to improve combustion efficiency. Unfortunately, current "low aromatic, low sulfur" diesel in California still contains 20 to 25% aromatics because the oil companies have been allowed waivers by the state to "reformulate" their diesel fuels and reduce emissions by adding "cetane enhancers" to lower emissions to levels equivalent to 10% aromatics. An engine running on 100% Biodiesel would have NO aromatic emissions and the Biodiesel would be much safer to store and handle. In addition, Biodiesel blends have reduced emissions of polyaromatic hydrocarbons, another group of potentially carcinogenic substances found in petroleum. As an oxygenated vegetable hydrocarbon, Biodiesel itself burns cleanly, but it also improves the efficiency of combustion in blends with petroleum fuel. As a result of cleaner emissions, there will be reduced air and water pollution from boats operated on Biodiesel blends. At a 20% Biodiesel blend, there will be a noticeable change in the odor and smoke in the exhaust. Older engines should also emit less soot under load and less carbon black during startup. Independent research programs in Europe and the U.S. have shown that Biodiesel in a 20 percent blend with petroleum diesel created a significant reduction in visible smoke and odor. The studies documented the reduction in hydrocarbons, carbon monoxide and particulate matter. Biodiesel is comprised of vegetable oil methyl esters, that is, they are hydrocarbon chains of the original vegetable oil that have been chemically split off from the naturally occurring "triglycerides". Biodiesel hydrocarbon chains are generally 16 to 20 carbons in length, and they are all oxygenated at one end, making the product an excellent fuel. As discussed below, several chemical properties of the Biodiesel allow it to burn cleanly and actually improve the combustion of petroleum diesel in blends. A recent (1997) survey of recreational boaters using Biodiesel on the San Francisco Bay confirmed these findings. Boaters buy Biodiesel for the benefits, and clean emissions from diesel engine exhaust is a major driving force in the marine market. From the survey results among 100 boaters using Biodiesel at various blends over the past 3 years, 98% reported an improvement in the exhaust odor (smells more like french fries), 91% reported a reduction in smoke, and 56% indicated a reduction in soot deposits on the transoms and decks of their boats. Biodiesel blends should have a beneficial impact on human health by reducing dangerous particulates and enhancing catalyst performance in vehicles. The National Biodiesel Board emphasizes the importance of reductions in EPA-regulated emissions by citing a 1993 study published in the New England Journal of Medicine which concluded that "fine particulate air pollution, or a more complex pollution mixture associated with fine particulate matter, contributes to excess mortality in certain U.S. cities." More recent work (1996-1998) confirmed that Biodiesel contributes to the reduction of heavier (longer chains, C13+) hydrocarbons as well as reducing carcinogenic polyaromatic hydrocarbons (PAHs). Several emissions reduction studies have been performed by the Southwest Research Institute (SWRI) over the past 5 years. In a 1994 study on light diesel trucks, Biodiesel in a 20% blend (B-20) was shown to reduce particulate matter (PM) by 14% in new engines. However, from our own from field observations with boats and test cars, Biodiesel appears to be even more effective in reducing smoke from the older engines typical of most recreational boats. The reduction in PM when B-20 is used is due to a reduction in insolubles (particles), generally composed of carbon soot. Catalytic converters (used in trucks and cars) can further contribute to the reduction in PM when B-20 is used. Subsequent SWRI studies were presented at a Biodiesel Emissions Testing Meeting in Seattle in 1996. The SWRI studies were conducted with the most efficient diesel engine produced by Cummins for pick up trucks: a 1995 5.9 Liter inline 6-cylinder, 4-stroke engine, with direct injection, a turbocharger and an intercooler. The rated horsepower of the test engine was 160 HP at 2500 rpm. Studies were conducted with and without the stock catalytic converter supplied with the engine (unfortunately not available for marine engines). The engine emissions tests were conducted under transient heavy duty loads to simulate road use. Several types of Biodiesel were tested, including Biodiesel derived from another oil crop, winter rapeseed. Rapeseed is grown extensively in Idaho, Canada and Europe to produce Canola. Biodiesel made from Canola has very similar properties to the Biodiesel made from soybean and other plant oils. In the Idaho study, the Biodiesel test fuels were (100%) rapeseed ethyl esters (REE), rapeseed methyl esters (RME), 50% blends of REE or RME, and 20% blends of REE or RME with petrodiesel. The reference fuel was conventional (Texas) No. 2 petrodiesel ("2D"), a low-sulfur diesel formulation mandated nationally by the EPA since 1993 . More recent work at the SWRI was presented at a Biodiesel Environmental Workshop in Washington, D.C. (June 4, 1998) by Christopher Sharp, the principal investigator. The newest work provides updates on the effects of Biodiesel on diesel engine exhaust emissions and performance. The 1998 SWRI report summarized emission studies on three diesel truck engines: a 1997 Cummins 14-L inline 6 cylinder engine, a 1997 Detroit Diesel 8.5-L incline 4 cylinder engine, and a 1995 Cummins 5.9-L inline 6 cyclinder engine. The test fuels were neat (100%) Biodiesel, the B-20 blend of biodiesel with diesel and a standard low-sulfur standard petroleum diesel fuel as a reference. Exhaust emissions were evaluated over a heavy duty transient cycle with hot and cold starts, with and without a catalyst in some cases. Total hydrocarbon emissions for the Cummins engines were reduced by 20% for the B-20 blend in the 1997 model and by 31% for the 1995 B-5.9 model. When the same engines were tested with 100% Biodiesel, there was a 94% drop in total hydrocarbons for the 1997 engine and a 72% drop for the 1995 engine relative to No. 2 low sulfur diesel fuel. The Detroit Diesel engine generated less emissions in general, with little change as the test fuel was switched to B-20, but an 82% drop when the test fuel was neat Biodiesel. Smoke (particulate material) and soot (unburned fuel and carbon residues) are of increasing concern to urban air quality problems that are causing a wide range of adverse health effects for their citizens, especially in terms of respiratory impairment and related illnesses. Boaters always complain of the smoke from their diesel engines as they motor back to port. They also resent the soot accumulation on the transoms and decks of their boats. The lack of heavy petroleum oil residues in the vegetable oil esters that are normally found in diesel fuel means that a boat engine operating with Biodiesel will have less smoke, and less soot produced from unburned fuel. Further, since the Biodiesel contains oxygen, there is an increased efficiency of combustion even for the petroleum fraction of the blend. The improved combustion efficiency lowers particulate material and unburned fuel emissions especially in older engines with direct fuel injection systems. In the 1996 study performed by the Southwest Research Institute, the effect of oxygen content (by percent oxygen) on the production of particulates smoke and soot was studied using the same Cummins diesel test engine cited above. The study established, for example, that a B-20 blend (approximately 2% oxygen for RME-20) reduces particulate soot by approximately 30% (from 0.06 for diesel no. 2 to 0.04 G/HP-Hr for rapeseed methyl ester at 20%). An earlier 1994 EPA Transient Cycle Emissions Test undertaken by the Southwest Research Institute compared emissions from an engine burning No. 2 low-sulfur diesel with those from an engine using the B-20 blend in combination with an oxidation catalyst. Compared with the low-sulfur No. 2 diesel, the B-20 blend with an oxidation catalyst reduced particulate matter by 45%, total hydrocarbons by 65%, and carbon monoxide by 41%. Pioneering studies performed by the Colorado Institute for Fuels and High Altitude Engine Research used 1991 model series engines. They reported a 13.7% drop in particulate matter and a 12.7% drop in total hydrocarbons when using a B-20 blend. In the updated 1998 report from the Southwest Research Institute studies, oxygen was shown to be the driving mechanism for soot reduction in truck engines operating on various blends of Biodiesel. The higher the oxygen content of the Biodiesel blend, the greater the reduction in soot emissions. However, not all the particulate emissions is fuel related. With the Cummins B-5.9 test engine, about 22% of the particulate emissions was unburned lubrication oil that did not change significantly when the fuels were switched to B-20 or neat Biodiesel. Carbon soot, in contrast, made up over 50% of the particulate emissions and this carbon soot component was reduced by 20% with the B-20 blend, and reduced with the neat Biodiesel by 66% in the Cummins B5.9 engine and by 71% with the Detroit Diesel engine. Carbon monoxide gas is a toxic byproduct of all hydrocarbon combustion that is also reduced by increasing the oxygen content of the fuel. More complete oxidation of the fuel results in more complete combustion to carbon dioxide rather than leading to the formation of carbon monoxide. In the 1998 report by the Southwest Research Institute on the effects of Biodiesel on truck engine exhaust emissions, the levels of carbon monoxide were shown to be reduced from 8% to 22% with a B-20 blend, depending on the type of engine. When the fuel was switched from low-sulfur petroleum diesel to neat Biodiesel, there was a 28% to 37% drop in the carbon monoxide emissions. Polyaromatic Hydrocarbon Emissions Polyaromatic hydrocarbons (PAHs) are a class of heavy oil petroleum hydrocarbons defined by their complex ring structures and unique qualities. They consist of multiple benzene ring structures that make them insoluble, slow to burn and carcinogenic. PAHs are regulated by the EPA in engine emissions. In the 1998 SWRI report, the Cummins N-14 engine had a 12% drop in PAH emissions when operating on B-20 blend relative to petrodiesel, and a 74% drop in PAHs when the fuel was switched to neat Biodiesel. The Detroit Diesel engine had a 29% reduction in PAHs operating on B-20 and a 68% reduction when operating on neat Biodiesel. These data suggest major gains in improving the air quality around diesel engines in vehicles and boats operating on Biodiesel. The nitrogen oxides result from the oxidation of atmospheric nitrogen at the high temperatures inside the combustion chamber of the engine, rather than resulting from a contaminant present in the fuel. Although nitrogen oxides (NOx) are considered a major contributor to ozone formation, they are also a reality of operating internal combustion engines. There are consistent reports of slight increases (several percent) in NOx emissions with Biodiesel blends that are attributable, in part, to the higher oxygen content of the fuel mixture. More oxygen and better combustion of the fuel also means more formation of NOx emissions with Biodiesel blends. In several research studies conducted since 1993 in the U.S. and Europe, EPA-regulated emissions from an unmodified engine operating on a 20% Biodiesel/80% petrodiesel blend were shown to be lower than those for petroleum diesel, except for NOx (nitrogen oxides) emissions, which can be 2-5% above baseline emissions. Some reductions in NOx emissions can be attained by retarding the timing of ignition and slowing the burn rate of the fuel in the combustion chamber. In the EPA Transient Cycle Emissions Test (Southwest Research Institute) study with the 1988 DDC 6V-92 engine, there was a 7% increase in NOx emissions that accompanied the 45% reduction in particulate matter and 65% reduction in hydrocarbons (in combination with an oxidation catalyst). Unfortunately, any improvements in NOx emissions are usually offset by increases in hydrocarbon, particulate material and carbon monoxide emissions caused by the mechanical adjustments to the engine. In the case of the Transient Cycle Emissions Test, a one-degree timing change in the diesel engine did result in a net reduction of NOx emissions by 2%, but at the expense of slightly less dramatic reductions in the particulate matter (reduced by 40%), hydrocarbons (reduced by 58%) and carbon monoxide (reduced by 34%). In Europe, the delays in engine ignition timing have been successfully combined with the use of catalytic converters to achieve similar reductions in both NOx emissions and hydrocarbon emissions from transit buses. In the 1996 Southwest Research Institute study cited above, the use of a catalytic converter improved the reduction of hydrocarbon emissions with a B-20 blend of rapeseed methyl esters from 29% (without converter) to 41% (with converter) for the Cummins test engine without any timing delays. NOx emissions were reduced 3%. Biodiesel Helps Reduce Greenhouse Gases Unlike other "clean fuels" such as compressed natural gas (CNG), Biodiesel and other biofuels are produced from renewable agricultural crops that assimilate carbon dioxide from the atmosphere to become plants and vegetable oil. The carbon dioxide released this year from burning vegetable oil Biodiesels, in effect, will be recaptured next year by crops growing in fields to produce more vegetable oil starting material. The U.S. is under considerable pressure from the international community (for example, at the December 1997 Kyoto Conference) to take seriously its efforts to reduce carbon dioxide, carbon monoxide and other greenhouse gases released, in part, by the combustion of fossil fuels in vehicles. While anthropogenic (man-made) CO2 production accounts for only about 4-5% of the net CO2 emissions, it is sufficient to have caused a net gain over the past 100 years. Fossil fuel combustion accounts for 70% of the total anthropogenic CO2 contribution. Supplementing our dwindling fossil fuel reserves with biomass-based fuels (Biodiesel, for petrodiesel; biomass-based alcohols or hydrogen for gasoline) helps reduce the accumulation of CO2. Positive Energy Balance for Solar Energy in Biodiesel Although it takes fossil energy to produce and transport biofuel, Biodiesel has a very favorable energy balance, especially relative to energy-negative ethanol from corn. Biodiesel production has positive energy balance ratios ranging from 2.5:1 (Institute for Local Self-Reliance) up to 7.4:1 in Europe, depending on oil crop and distance required to transport the raw materials. LOWER IMPACT ON MARINE ENVIRONMENT Water pollution should also be reduced by using Biodiesel in boat engines since there will be more efficient burning of the fuel mixture, less carbon (soot) accumulation and particulate (smoke) emissions. Faster starting and smoother operation also should reduce the discharge of unburned fuel. Any accidental discharges of small amounts of Biodiesel should have relatively little impact on the environment compared to petroleum diesel, which contains more toxic and more water-soluble aromatics. Nonetheless, the methyl esters could still cause harm. Comparatively Low Toxicity to Marine Plants and Animals From 1994 through 1996, CytoCulture conducted a series of tests in collaboration with the California Department of Fish & Game (Office of Oil Spill Prevention & Response) to document the impact of vegetable methyl esters on various native species of marsh plants and marine organisms. Because larval forms of fish and shell fish are much more sensitive than the adult forms, all of the marine toxicity studies were performed with larvae of established test species. The studies indicated that the Biodiesel, while not completely harmless to the larvae of crustacea and fish, is much less toxic than petroleum fuels and crude oil. In research conducted for CytoCulture in 1994, the LC50 (concentration required to kill 50% of the population) for larval test fish (Menidia Beryllina) exposed to soy methyl ester Biodiesel was 578 ppm relative to an LC50 of 27 ppm for reference fuel oil. In larval shrimp (Mysidopsis Bahia) toxicity assays, the LC50 for the soy methyl ester Biodiesel was 122 ppm compared to the LC50 of 2.9 ppm for the reference fuel oil. In 1996, follow-up acute toxicity bioassays were performed by a different laboratory on recycled cooking oil methyl esters for CytoCulture using the same protocol and the same two test species. The recycled cooking oil Biodiesel had an average LC50 of 736 ppm in the Menidia fish larvae tests compared to the average LC50 of 39 ppm for the reference fuel oil. In the Mysidopsis toxicity bioassay, the recycled cooking oil Biodiesel had an LC50 of 124 ppm compared to the LC50 of 5.9 for the reference fuel oil. In subsequent gas chromatography studies on the solubility of Biodiesel methyl esters, it was determined that much of the apparent toxicity observed in abalone, shrimp and fish larvae was due to suffocation or coating of exposed gills as the tiny larvae swam into dispersed globules of the methyl esters. The concentrations for the reported LC50’s exceed the saturation concentration for Biodiesel in seawater, further indicating that the observed marine toxicity is probably due to the formation of globules rather than due to a true chemical toxicity of dissolved-phase product. In other words, the Biodiesel methyl esters have very low solubility in water (7 ppm in San Francisco Bay seawater) compared to petroleum diesel that contains benzene, toluene, xylene and other more water-soluble, highly toxic compounds. However, when the Biodiesel is vigorously blended into water (as required by the EPA test protocols), the methyl esters form a temporary emulsion of tiny droplets that appear to be harmful to the swimming test larvae. In earlier investigations of Biodiesel, aquatic toxicity studies conducted in 1993 by CH2M Hill in Oregon indicated that rapeseed methyl esters had an LC-50 of 23 ppm for Daphnia Magna (shrimp larvae) relative to diesel fuel which had an LC50 of 1.43 ppm, a 15-fold difference in apparent toxicity. Acute bioassays of bluegill adult fish conducted at the University of Tennessee in 1996 showed that fish dosed with engine exhaust from a 110-HP Volvo marine diesel engine experienced mean mortality values of 25.9% and 22.1% for biodiesel and petrodiesel, respectively. The investigators commented that the petrodiesel dispersion in the engine’s wet exhaust tended to settle out more than the biodiesel, and therefore might have appeared to have less impact on the fish than the biodiesel droplets which remained suspended in the water. However, the study also noted that the production of carbon monoxide and soot was reduced by using Biodiesel. A major health and environmental concern with engine exhaust is the emission of polyaromatic hydrocarbons from burning fuel and engine oil. Polyaromatic hydrocarbons (PAHs) found in smoke and soot are complex, multi-ring compounds that can be carcinogenic and toxic. Water dosed with biodiesel exhaust at a 50% engine load had non-detectable levels of polyaromatic hydrocarbons (PAHs are complex multi-ring compounds in petroleum that can be carcinogenic), while the exhaust from the engine operating with petroleum diesel had anthracene and naphthalene, both carcinogenic PAHs. Low Solubility and High Biodegradation Rate for Biodiesel in Bay Water Biodiesel methyl esters are actually quite insoluble in fresh or sea water, with a saturation concentration of 7 ppm (sea) and 14 ppm (fresh) at 17 Deg. C, whereas petroleum diesel can partition aromatics into water in concentrations of hundreds of ppm. The dissolved phase of the Biodiesel methyl esters was shown to breakdown by the biodegradation action of naturally occurring bacteria present in San Francisco Bay sea water. The half-life for the biodegradation of the vegetable methyl esters in agitated SF Bay water was less than 4 days at 17 Deg. C., about twice as fast as petroleum diesel (reported by others). In the environmental remediation field, CytoCulture developed an oil spill cleanup solvent (CytoSol Biosolvent) based on vegetable oil methyl esters similar to Biodiesel. The CytoSol Biosolvent has been licensed by the California Department of Fish & Game as a "shoreline cleaning agent" to extract crude oil from shorelines and marshes after a spill. The acceptance of this product by the state for application to shorelines emphasizes the low toxicity and low water solubility of the esters. See the CytoCulture web site for more background on the CytoSol Process cleanup technology. Biodegradability of Biodiesel in the Aquatic Environment A study conducted at the University of Idaho in 1995 determined that rapeseed Biodiesel would biodegrade about twice as fast as petroleum diesel using a standard EPA test protocol based on carbon dioxide evolution and gas chromatography. Further, the Biodiesel was shown to enhance the biodegradation rate for diesel fuel in a blend. As was also confirmed at CytoCulture, hydrocarbon-degrading bacteria can metabolize both the Biodiesel and petroleum diesel at the same time. Because Biodiesel is a simple, straight carbon chain with two oxygens at one end (mono-alkyl ester), it is more readily metabolized by bacteria that normally break down fats and oils in the environment. The petroleum diesel hydrocarbons lack oxygen, and represent a very complex mixture of hydrocarbons with multiple double bonds, and many other branched, cyclic and cross linked chains. The more complex chemical strucutures of diesel hydrocarbons makes them more difficult to biodegrade, and in many cases, toxic. The biodegradation rate of rapeseed biodiesel in shake flasks with fresh water was found to be comparable to dextrose (a test sugar) and about twice as fast as for petroleum diesel. In the Idaho study (Peterson, Reece, et al., 1996), the rapeseed esters were degraded by 95% at the end of 23 days where as the diesel fuel in this test was only about 40% degraded after 23 days. Spills of Biodiesel Can Still Harm the Environment For the boating environment, Biodiesel should have less impact to aquatic and marine organisms than petroleum diesel if accidentally spilled or inadvertently discharged over the side. However, the US EPA still considers spills of animal fats and vegetable oils harmful to the environment. In an October, 1997 ruling under the Clean Water Act, as amended by the Oil Pollution Act of 1990, vegetable oils are considered "oil" like petroleum. (In France, Biodiesel is classified as food for transportation purposes.) Spilling Biodiesel into the water would be as illegal as discharging petroleum fuels overboard. Waterfowl and other birds, mammals and fish that get coated with vegetable oils could die from hypothermia or illness, or fall victim to predators. Even though the Biodiesel is relatively non-toxic and less viscous than vegetable oil, it can still have a serious impact on marine and aquatic organisms in the event of a big spill. We recommend that the Biodiesel always be handled like any other fuel to avoid contamination of our bays and waterways, and that boaters obey all laws governing the handling of engine fuels and oils. Mechanical Advantages to Using Biodiesel with Reformulated "Low Sulfur, Low Aromatics" Diesel Biodiesel methyl esters improve the lubrication properties ("lubricity") of the diesel fuel blend. Long term engine wear studies have been conducted in Europe and in the US. Porsche (Germany) determined that neat (100%) Biodiesel reduced long term engine wear in test diesel engines to less than half of what was observed in engines running on current low sulfur diesel fuel. Lubricity properties of fuel are important for reducing friction wear in engine components normally lubricated by the fuel rather than crankcase oil. When the California Air Resources Board (CARB) mandated stricter laws than the Federal requirements for reformulating "low sulfur, low aromatic" diesel fuel in 1993, the result was a decrease in the lubricity of that fuel. The reduction in aromatics at that time also changed the elastomeric properties of the fuel resulting in the shrinking of gaskets, O-rings and seals in older engines. The mechanical wear and fuel leaks caused so many problems (e.g., expensive rebuilds of fuel pumps) that California truckers held a one day strike in December, 1993 to protest the new fuel laws. Since then, truckers, boaters and other operators of diesel engines have turned to a variety of petroleum additives (in extreme cases, transmission fluid) in an attempt to protect their engines from excessive wear and gasket leaks associated with the new "CARB low sulfur/low aromatics" diesel fuel. More than 100 Biodiesel demonstrations, with over 10 million road miles in trucks, have confirmed the performance benefits of this fuel additive for emissions and mechanical lubricity. No adverse durability or engine wear problems were found; in fact, in road tests with heavy duty truck engines, engine wear was significantly decreased after running 100,000 miles on blends of Biodiesel (University of Idaho studies). Biodiesel has been studied extensively in Europe and the U.S. for its effect on long term engine wear, particularly with respect to those components normally lubricated by the fuel itself. Fuel pumps and injector pumps depend on the operating fuel for lubrication of moving parts and shaft bearings. Initial work on the lubricity of Biodiesel, performed by Mark-IV Group and the Southwest Research Institute in 1994, established a clear advantage to blending Biodiesel with petrodiesel to achieve superior lubrication. Lubricity properties are measured at the Southwest Research Institute (SWRI) by a "Ball On Cylinder Lubricity Evaluator" (BOCLE) machine to measure metal to metal hydrodynamic wear simulating rotating shafts and bearings. A static steel ball is loaded onto the edge of a rotating disc and the diameter of the subsequent scar on the ball is measured (similar reciprocating machines exist in Europe to measure scar on a steel ball, and newer versions have been developed in America to improve lubricity measurements). The BOCLE test does not measure adhesive friction wear. Tests run by Exxon showed that, compared to reference diesel fuel in 1993, a 20% blend of Biodiesel had significant, quantifiable improvements in reducing wear (193 micron scar for B-20 vs. 492 micron scar for petrodiesel) and friction (0.13 micron scar for B-20 vs. 0.24 micron for petrodiesel) while improving film coating ability of the blend (93% film with the B-20 vs. 32% film with the petrodiesel). The B-20 blend compared favorably for lubricity results against Exxon’s own lubricity additive. The SWRI results for the BOCLE tests confirmed the earlier Exxon study results. Low sulfur, low aromatic ("CARB") diesel was compared to various blends of Biodiesel (soy methyl esters). Data were presented in values of grams of weight added to the apparatus before failure of the fuel to adequately lubricate the metal. The higher the weight the ball could support, the better the lubricity of the fuel. Neat petrodiesel (low aromatic CARB) had a BOCLE result of 3,500 grams, whereas the neat Biodiesel had a BOCLE result almost twice as high at 6,100 grams. The B-20 blend had a BOCLE result of 4,100 grams, close to the value for pre-1993 (high sulfur, high aromatic) petrodiesel fuel. In concentrations below 5%, the Biodiesel had no measurable effect on the lubricity of petrodiesel. Follow up BOCLE studies at SWRI in 1996 concluded that Biodiesel methyl esters had even better lubricity properties than previously reported. The Biodiesel (RME) had a BOCLE value of 7,000 grams vs. 4,250 for low sulfur diesel (not CARB diesel), and the B-20 blend had a BOCLE value of 4,600 grams. Scar wear diameters were also encouraging, with a 405 micron scar reported for petrodiesel vs. a 190 micron friction scar for the B-20 blend. Subsequent field studies on light duty truck engines (5.9L Cummins diesel at the University of Idaho) have corroborated these results by finding an "absence of wear" and friction scars on engines broken down for inspection after a 100,000 mile road test running on 28% Biodiesel. In a University of Idaho durability test (1,000 hour tests on small diesel engines), it was found that methyl ester Biodiesel was equivalent to no. 2 diesel on the basis of long term engine performance and wear. The primary factors evaluated in that study were engine brake power and torque, injector tip coking (carbon deposition), and engine component wear based on oil analysis. In house monitoring over the past 5 years of our "Biofuel Test Vehicles" (a Mercedes Benz 300TD diesel station wagon and a 1985 BMW 524-Diesel) at CytoCulture has shown no evidence of unusual wear or polymerization of engine crankcase oil (analysis performed by Herguth Laboratories, Vallejo, CA) after more than 40,000 miles of operation on 30-100% blends with Biodiesel. Relative to petroleum diesel no. 2, Biodiesel has a slightly lower heat of combustion on account of its oxygen content (petroleum diesel hydrocarbons are not oxygenated). The heat of combustion for soy methyl esters is 128,000 BTU (British Thermal Units) per gallon vs. 130,500 BTU/gal. for petrodiesel. In the Southwest Research Institute study (1996), the heat of combustion for rapeseed biodiesel in blends were compared with petrodiesel. Petrodiesel had 18,400 BTU/lb., neat biodiesel had 16,200 BTU/lb. (88%) and a 20% blend of rapeseed methyl ester biodiesel had 17,900 BTU/lb. (97%). However, with the added oxygen, the net combustion efficiency for the blended fuel is increased, which should compensate for the slight drop in BTU content. The differences would be most noticed at low rpm and high engine load when the engine would most benefit from more oxygen. Studies conducted in the U.S. and Europe generally indicate that blends of Biodiesel and petrodiesel result in small decreases in overall power output of engines. Only two studies have been conducted with marine engines, one by a German scientist (Dr. Claus Breuer) at the Technical University in Hannover (Ph.D. thesis in 1994) and the other by Alvin Womac’s group at the Department of Agricultural Engineering at the University of Tennessee. The German study involved a Deutz 4 cylinder marine diesel engine (direct injection) found on fishing boats in Europe and the Tennessee study evaluated a 110 HP Volvo marine diesel engine, also used in work boats and fishing boats. Volvo also makes smaller single and double cylinder diesel engines for recreational sailboats. The German study confirmed similar results obtained by Mercedes Benz showing that the maximal torque curve for an engine under load remains essentially unchanged for rapeseed methyl esters relative to pure petrodiesel. Despite the lower volumetric heating value and the consequent lower maximum power output of Biodiesel, the practical results are roughly the same. At a 20% blend, there would probably be no noticeable difference in power output. Good performance in fuel combustion with Biodiesel and its blends resulted in a smooth running engine. In the Volvo marine diesel engine study in Tennessee (110-HP, 2.39 L, 4-cylinder, direct injection engine), a tractor dynamometer was used to measure power outputs under selected loads through an engine-mounted reverse drive gear. Exhaust emissions were also tested along with fuel consumption tests under various loads. The conclusions of these tests were that power produced from 100% soy methyl ester Biodiesel was from 2 to 7 percent less than produced from petrodiesel, depending on the load-speed point. However, at or near maximum throttle (3,800 rpm), the two fuels performed the same. Interestingly, at the lowest engine speed (1855 rpm) at full throttle under heavier load, there was a 13% increase in power with Biodiesel as compared to petrodiesel. The Tennessee study indicated that using 100% Biodiesel in marine direct-injection diesel engines, with design and construction similar to the Volvo test engine, could be recommended without any significant, noticeable differences in operation, power performance and fuel usage. In the 1998 study at the Southwest Research Institute on Biodiesel effects on diesel engine performance, engine power in the 1997 Cummings truck engine operating on the B-20 blend was at 98.5% of the power attained with low sulfur No. 2 diesel. At 100% Biodiesel, the engine generated 92% of the power. For a Detroit Diesel truck engine (1997), the power was 98% with the B-20 and 92% with the neat Biodiesel. Biodiesels are mono-alkyl esters containing approximately 10% oxygen by weight. The oxygen improves the efficiency of combustion, but it takes up space in the blend and therefore slightly increases the apparent fuel consumption rate observed while operating an engine with Biodiesel. In the Southwest Research Institute study (1996), the fuel consumption was found to increase by only 2% for a B-20 blend with methyl esters, and by 14% when methyl ester Biodiesel was used at 100% in the Cummins test engine operated under transient heavy loads. The brake-specific fuel consumption was 0.43 lb./HP-Hr for regular petrodiesel no. 2, 0.44 lb./HP-Hr for the B-20 blend, and was 0.50 lb./HP-Hr for the neat RME Biodiesel. In testing Biodiesel in the CytoCulture Mercedes Benz diesel station wagon over the past 4 years, there was about a 15% net decline in the mileage obtained using neat Biodiesel vs. petrodiesel. No change in power, acceleration or engine temperature was observed, but the engine was quieter and smoother at idle when fueled with Biodiesel. At a 20% blend with petroleum diesel, the fuel consumption differences are practically unnoticeable. These local observations were confirmed by the 1998 engine performance studies at the Southwest Research Institute. Fuel consumption in a 1995 Cummings B-5.9 truck engine increased by 9% with the B-20 blend, and by 18% with the neat Biodiesel. Better fuel economy was noted for a 1997 Cummings N-14 truck engine with a 3% drop in fuel consumption using B-20 and a 13% increase with the neat Biodiesel. Engine Seals, Gaskets and Hoses The oxygenated methyl esters of vegetable oil cause Biodiesel to have surprisingly strong solvent properties with respect to natural rubber and several soft plastics. As a result, old rubber fuel lines and some seals or gaskets on fuel tanks may slowly deteriorate in the presence of higher concentrations of Biodiesel. Fortunately, few of these solvent effects are noticed at a B-20 blend, and most of the problems associated with the solvent effects occurred with boats using 100% neat Biodiesel. When fuel lines or gaskets are affected, they usually get sticky over time and soften or swell, causing fuel to drip from connections. In one case, the rubber fuel line between the primary filter and the fuel pump on a Yanmar sailboat engine became tacky, but did not leak, after 4 years of operating on 100% Biodiesel. The best solution is to replace affected lines and gaskets with modern synthetic hoses and seals. Conventional US Coast Guard approved fuel lines are resistant to Biodiesel (neat) and proven in sailboat testing over the past 3 years. In California, an approved fuel hose readily available in marine stores is: "Trident Barrier Fuel Hose, USCG Approved Type A-1, SAE J1527 (2/93)" In bench top studies conducted at CytoCulture, the Trident hose proved to be resistant to neat Biodiesel over a period of months, although the hose did absorb Biodiesel and swell slightly (tightens under hose clamps). With 20% blends, there have been no reports of any problems with these new fuel hoses. Even at 100% Biodiesel, we have observed only minor swelling on the Trident Barrier fuel hoses used on test engines operating on neat Biodiesel for several years. Studies conducted for the National Biodiesel Board on the materials compatibility of Biodiesel concluded that the only hose and gasket material that was truly resistant to the solvent effects of methyl esters was Viton. Viton fuel hoses (Goodyear) can be special ordered for boats (usually expensive at over $5.00/ft for 5/16" line), but we know of only one boat in the San Francisco area that converted to Viton fuel lines as a precaution. In CytoCulture’s 1997 survey of 100 boaters using Biodiesel in the San Francisco Bay area, 2% of the respondents had trouble with drips caused by swelling gaskets and seals, usually at the fuel filter. Again, replacing these gaskets with modern synthetic materials appeared to solve the problem. Raycor filters, for example, have functioned normally with 100% Biodiesel and have had no gasket problems in engines operated with neat Biodiesel over the past 4 years. (The 1997 boater survey is on the CytoCulture web site). In the survey, 5% of the boaters reported minor problems with the Biodiesel if they spilled it on decks, on their engine or into their bilges. The solvent properties of the esters in Biodiesel can loosen old paint on engines or on painted surfaces in the bilge. Besides staining raw wood surfaces, the Biodiesel is particularly harmful to teak decks with polysulfide seams (use extra caution when filling tanks via deck ports). The Biodiesel could also harm rubber engine mounts if it were spilled and not cleaned up immediately. Use paper towels or absorbant pads to remove spilled Biodiesel and then clean the surfaces thoroughly with warm soapy water. Warranties and Engine Manufacturer Endorsements Marine diesel engine manufacturers in United States, Europe and Japan have all recognized the growing role of Biodiesel as a viable fuel additive, and in most cases, as a complete alternative fuel (100%). Two of the sponsors of the SUNRIDER expedition of 1992-1994 were the marine diesel engine manufacturers: Mercruiser (inboard/outboard diesel engine) and Yanmar (outboard diesel engines), endorsing Biodiesel as a suitable alternative fuel to power Bryan Peterson’s 28-ft inflatable Zodiac boat around the world. This 35,000 mile adventure remains the most famous and most publicized demonstration of using Biodiesel in marine engines. Over 18,000 gallons of donated soy methyl ester Biodiesel was provided to SUNRIDER at various destination ports and rendezvous locations (including a mid-ocean fuel transfer from a ship). Bryan started out from Pier 39 in San Francisco in 1992 and returned under the Golden Gate bridge on September 8, 1994, powered by 100% soybean Biodiesel. Brian’s last 100 gallons of Biodiesel were donated by CytoCulture when he stopped in Santa Cruz on the final leg up the coast of California. At that point, he remarked, "The Biodiesel works….No problems." Engine manufacturers in Europe have a long history of supporting the Biodiesel movement, and those that produce marine engines continue to endorse the alternative fuel use in their equipment. Some manufacturers warranty their marine engines for use with 100% Biodiesel for late models or for older engines retrofitted with newer synthetic hoses and gaskets that proved more resistant to the pure methyl esters over extended periods of time. Some prefer to warranty Biodiesel engines on a case by case basis. In the U.S., diesel engine manufacturers generally stand by their warranties as long as the fuel used in their engines meet the ASTM D-975 standards defining fuel for compression ignition engines. All of the B-20 blends of Biodiesel produced in America meet the ASTM D-975 specifications. Contact your engine manufacturer for updates on their acceptance of B-20 blend as an acceptable fuel within the scope of their warranties. SAFETY AND AESTHETIC ADVANTAGES OF BIODIESEL Boaters can appreciate the user friendliness of handling Biodiesel in their boats. The product has no noxious odors and is considered as harmless to handle as salad oil, but we always encourage safety precautions to avoid splashing it in your eyes, on your clothes, on your boat or into the water. The product smells and feels like cooking oil. In an early study sponsored by the National Biodiesel Board (1993), the product had less toxicity in animal testing than table salt (grams per kg body weight). No Noxious or Carcinogenic Fumes Biodiesel vegetable oil methyl esters contain no volatile organic compounds that would give rise to any poisonous or noxious fumes. The Biodiesel does not contain any aromatic hydrocarbons (benzene, toluene, xylene) or chlorinated hydrocarbons. There is no lead or sulfur to react and release harmful or corrosive gases. However, in blends with petrodiesel there will continue to be significant fumes released by the benzene and other aromatics present in the petroleum fraction (80%) of the blend. No Risk of Explosion from Vapors Since the Biodiesel has no volatile components (vapor pressure of less than 1 mm Hg) and a high flash point (typically over 360 Deg. F), the product poses no risk of explosion caused by fumes accumulated below deck. The only significant fire risk would be from the spontaneous combustion of rags and paper towels soaked in Biodiesel and stored in an area with low ventilation, or high temperatures (like the inside of an engine room). STORAGE CONDITIONS FOR BIODIESEL Biodiesel can be stored for long periods of time in closed containers with little head space. The containers should be protected from weather, direct sunlight and low temperatures. Avoid long term storage in partially filled containers, particularly in damp locations like dock boxes. Condensation in the container can contribute to the long term deterioration of the petroleum diesel or biodiesel (see below). Low temperatures can cause the Biodiesel to gel, but the Biodiesel will quickly liquefy again as it warms up. In cold weather (near or below freezing), additives can be used to prevent gelation (fuel additives for diesel fuel used in cold weather are available from Exxon, Hammond, and other manufacturers). Fuel tanks should be kept as filled as possible (regardless of whether they contain Biodiesel), particularly during rainy winter months or periods of inactivity, to minimize the condensation of moisture. Condensed moisture accumulates as water in the bottom of your tank and can contribute to the corrosion of metal fuel tanks, especially with petroleum diesel that also contains sulfur. The condensed water in the fuel tank can also support the growth of bacteria and mold that use the diesel and Biodiesel hydrocarbons as a food source. These hydrocarbon-degrading bacteria and molds will grow as a film or slime in the tank and accumulate as sediment over long periods of time. These hydrocarbon-degrading microbes are frequently referred to incorrectly as "algae" in advertisements for fuel treatments, perhaps because the colonies often have a reddish orange color and tend to form mats. Petroleum diesel and Biodiesel are both susceptible to growing microbes when water is present in the fuel, but the solvent action of the Biodiesel can also cause microbial slime to detach from the inside of the tank. The accumulation of the newly released slime and sediment can be very dangerous if it clogs the fuel filters and causes the engine to suddenly stop. It is very important to monitor the filters on a diesel engine that has been switched over to Biodiesel, particularly if the tank is old and has not been cleaned. Biocides are available at marine stores to treat diesel fuels suspected of having microbial growth. The biocides are chemicals that kill bacteria and molds growing in fuel tanks without interfering with the combustion of the fuel or the operation of the engine. Used in very dilute concentrations, the biocides can inhibit the growth of microbes over long periods of time. These products are very toxic and should be used only as directed by the manufacturers. Precautions should be taken to avoid any contact with the products (wear gloves and eye protection) and to prevent any spills or drips. It is important to remember that the biocides may kill the microbes, but they do not remove the accumulated sediment, so expect to replace fuel filters often as the debris is drawn from the tank. In some cases, it may be necessary to have the fuel filtered and the fuel tank cleaned by a professional fuel filtering service. The microbial slime and sediment problem seems to worsen for boats that are used infrequently since the inactivity allows the microbes to accumulate in stable colonies. When the boat is used again, the slime and sediment can break loose and accumulate in the fuel filters. Accumulated sediment in fuel filters can then interrupt the flow of fuel and shut down the engine, potentially with disastrous consequences. In recent years, several sailboats have washed up on beaches on account of clogged fuel filters with ordinary petroleum diesel caused by the sudden agitation of tank sediments when the boat encountered rough seas off shore. As mentioned earlier, the addition of Biodiesel to a dirty fuel tank can accelerate the release of accumulated slime. When the boat is then used after sitting idle for a long period of time, the newly suspended sediment can accumulate and potentially clog the fuel filters. We urge all boaters to check their fuel filters often and be prepared to change them after they introduce Biodiesel to an older fuel tank that may have accumulated slime and sediment. Biodiesel Producers in America Biodiesel (vegetable oil methyl esters) are produced in the United States as a feedstock for other consumer products (e.g., Dawn detergent), or as an end product biofuel or solvent. These products meet the draft specifications developed by the National Biodiesel Board, and they meet most or all of the European specifications for Biodiesel. These specifications assume the Biodiesel is used neat (100%) as an alternative fuel. At least 7 producers are currently supplying the American market with marine biodiesel from soybean methyl esters or recycled cooking oil methyl esters. Producers using only virgin soybean oil to make Biodiesel include: Ag Environmental Products (AEP): "SoyGold" 9804 Pflumm Road, Lenexa, KS 66215 Tel. 800-599-9209 Fax 913-599-2121 Web site: www.soygold.com SoyGold may be purchased from several fuel docks in Southern California. Check the SoyGold web site for fuel dock locations. Producers or distributors selling Biodiesel made from virgin soybean oil and/or recycled cooking oil: Columbus Foods, Inc.: "Biodiesel" 800 North Albany, Chicago, IL 60622 Tel. 800-322-6457 Fax 773-265-6985 Web site: www.columbusfoods.com NOPEC Corporation: "BioBooster" 1316 G. Jenkins Blvd., Lakeland, FL 33815 Tel. 888-296-6732 Fax 941-683-1058 Web site: www.nopec.com World Energy Alternatives (former Twin Rivers Technologies) 1 Broadway Suite 600, Cambridge, MA 02142 Tel. 617-621-1522 Fax 617-621-1523 Web site: www.worldenergy.net Producers using recycled cooking oil exclusively to make Biodiesel: Pacific Biodiesel, Inc.: "Biodiesel" 285 Hukilike Street, B-103, Kahului, Maui, HI 96732 Tel. 808-871-6624 Fax 808-871-5631 Web site: www.biodiesel.com Griffin Industries / PMC Marketing Group: "Bio G-3000" 4221 Alexandria Pike, Cold Spring, KY 41076-1897 Tel. 703-256-4497 Fax 703-256-8585 Web site: www.griffinind.com CytoCulture is a regional distributor of marine biodiesels from recycled cooking oil and a contract supplier of B-20 for fleets. Double Environmental Benefit with Biodiesel from The newer Biodiesel products (introduced in 1996-1997) produced from recycled cooking oil offer a "double environmental benefit" in being both renewable and recycled bioenergy products. Waste cooking oil collected from restaurants (e.g., fryer oil for fast foods, french fries, Chinese food, donuts, etc.) can be processed into Biodiesel methyl esters suitable for use as a fuel additive. In terms of performance, handling and marine toxicity, these products are virtually identical to the Biodiesel methyl esters produced from virgin soy bean oil in the U.S. The recycled products do have a darker, amber color (oxidized carotene pigments), but the trace pigment concentrations have never been associated with any engine performance or toxicity effects. In Europe, extensive testing of Biodiesel produced from recycled cooking oils has confirmed that their engine performance and exhaust emission properties are nearly identical to those of methyl esters produced from virgin vegetable (rapeseed) oil. The Europeans have built several new transesterification plants recently to meet the increasing demand for Biodiesel by processing waste cooking oil into methyl ester fuel. All four of the American recycled vegetable oil methyl ester products meet all specifications for American Society for Testing and Materials (ASTM) provisional standards for Biodiesel. All four recycled products were also shown to pass most European Union and German (final draft) specifications for Biodiesel. All of these test protocols are predicated on using the Biodiesel as a neat (100%) alternative fuel, rather than as blends with petrodiesel. In B-20 blends (20% Biodiesel / 80% CARB diesel), Biodiesel derived from both virgin soybean oil and recycled cooking oil meet ASTM D-975 standards defining "diesel fuel" used in compression ignition engines (See ASTM D-975 test results in appendix). The State of California uses the ASTM D-975 standard to enforce statutory laws (CA Department of Food & Agriculture, Division of Measurements) defining what is sold as fuel for diesel engines in California. Neat Biodiesel (100%) does not qualify as a diesel fuel in California on account of the product’s increased viscosity, and the fact that the product is not volatile (carbon residue tests and distillation curve data fail specifications). ASTM D-975 studies conducted with Herguth Laboratories in California indicated that neither the recycled cooking oil Biodiesel (NOPEC) nor virgin soybean oil Biodiesel (SoyGold from a SF fuel dock) could meet the ASTM D-975 specifications of a satisfactory diesel fuel. However, the B-20 blends met all D-975 specifications. From an environmental resource conservation point of view, building small regional plants to produce Biodiesel from recycled cooking oil offers a tremendous advantage in energy and resource savings. The ideal scenario for producing an alternative fuel or additive would be to have a local plant process local waste oil for use in a local market. This scenario is already a reality in Florida and Hawaii, and will soon be repeated in other regions of the country where the combination of local market demand (e.g., boaters, government vehicles, etc.) and abundant feedstock (used cooking oil) allow the concept to make good economic sense. In Idaho, at an enormous french fry processing plant, semi-tractor trucks haul potatoes and frozen french fries hundreds of miles using a blend of petrodiesel and Biodiesel made from recycled fry oil at the plant in collaboration with the University of Idaho. An obvious and important advantage of employing a local plant to produce Biodiesel from local, low cost feedstocks for local market consumption is lower cost. The recycled cooking oil Biodiesels are already sold locally at prices significantly lower than the Biodiesel products made from virgin soybean oil in the Midwest (or distributed on the East coast). Presumably, smaller plants could be built in other areas of the country like California to produce methyl esters with lower transportation and feedstock costs, thereby helping to boost the market share for Biodiesel among boaters and other potential users. The recent success of the "Veggie Van" is a prime example of how even a mobile transesterification plant can generate adequate quality Biodiesel on the road. In 1997-1998, Josh and Kaia Tickell crossed the country with their Biodiesel-powered "Veggie Van" processing used cooking oil into fuel by towing a small trailer-mounted system they built themselves. See their web site (www.veggievan.org) and read their book to learn more about their experience with Biodiesel: From the Fryer to the Fuel Tank: How to Make Cheap, Clean Fuel From Free Vegetable Oil. CytoCulture provided the Veggie Van with Biodiesel during their stay in California in the summer of 1998. Biodiesel as a "Mainstream" Alternative Fuel Global resources of vegetable oils and fats amount to some 62 million tons per year, which is small compared to the annual global petroleum consumption of 3,300 million metric tons (1995). Available vegetable oil resources could cover less than 2% of the present petroleum consumption, but it already has displaced 5% of the petroleum diesel consumed in France. In Germany last year, Biodiesel use was increasing faster than the plants could produce it, but even then, with virgin as well as recycled feedstocks, the maximum production of Biodiesel based on current agricultural practices could replace only about 6% of the total petroleum diesel used today. Tourist boats on lakes in Germany and Austria routinely run on Biodiesel. On the Bodensee (Lake Constance) that serves as a drinking water reservoir, some vessels have been operating on neat (100%) Biodiesel for over 3 years. The intent of all Biodiesel programs is not to replace petroleum entirely as a fuel, of course, but to displace it in key applications where the environmental impacts of diesel fuel are most threatening and can be easily muted by replacing the fuel with Biodiesel or a blend of Biodiesel. Recreational sailboats are a prime target for using Biodiesel since they operate in Bays, estuaries, lakes and rivers, and sailors tend to be very sensitive to the aesthetic and environmental concerns of operating engines. Furthermore, since sailboaters use relatively low volumes of fuel per year, the high cost of Biodiesel is less inhibiting than it would be for power boat and ferry operators or commercial fishermen. As state and Federal laws evolve over the next few years to accommodate Biodiesel as an approved alternative diesel fuel (with full fledged ASTM specifications), more plants will be constructed to process Biodiesel from recycled cooking oils in more coastal areas of the country (e.g., California). Lower production costs (local feedstock, local market) will help lower the price of Biodiesel and increase its use in boats and vehicles. As a result of recent legislation (fall, 1998), the US EPA and the Department of Energy now designate the B-20 (20% Biodiesel) blend as an official "alternative fuel" in the Alternative Fuel Transportation Program regulations. The legislation allows government fleets to earn equivalent "credits" for using the Biodiesel blend in conventional diesel-powered vehicles instead of having to buy more expensive, specialized "multi-fuel vehicles" to remain in compliance with the Energy Policy Act. The new policy is being drafted and should be published in the Federal Register by late spring, 1999. The new legislation was passed after recognition that "there is a developing market for biodiesel fuels with fuel producers prepared to meet the needs of this market". The Senate legislation, S.1141, will allow Biodiesel to compete on the same playing field as other alternative fuels and will expand clean-air options for city fleet managers, as well as encourage opportunities for boaters, farmers and other prospective users without additional government spending, federal mandates or subsidies. This legislation has led government fleets in California to consider buying conventional diesel-powered transit buses and work vehicles to operate on the B-20 blend and receive their Energy Policy Act "fuel for credit" in lieu of buying the multi-fuel vehicles. The capital expenditure and vehicle maintenance savings will more than offset the $0.60-$0.70 per gallon increase for the B-20 blend relative to the regular petroleum diesel. CytoCulture recently received the first contract to provide the B-20 Biodiesel blend to a government fleet in California at the Davis campus of the University of California. This is the first fleet in California and the first University fleet in America to convert entirely to operating on the B-20 blend of Biodiesel. As more fleet opportunities develop for the sale of the B-20 blend, and Biodiesel becomes more of a mainstream alternative fuel, there should be greater incentive to construct transesterification plants on the west coast to convert local recycled cooking oil into Biodiesel for local markets and at lower costs. In another development favoring Biodiesel use in California, the state senate is working this spring on the passage of legislation to exempt Biodiesel blends (B-20) from state excise fuel tax. The fuel tax on diesel fuel ($0.18 per gallon) is now applied to the entire volume of a B-20 blend sold as a blend. As a result, consumers and fleets end up paying 18 cents a gallon fuel tax in addition to sales tax (7.25-8.25%) on the Biodiesel portion of the B-20 blend. Under the proposed legislation of Assembly Bill 448, both Biodiesel and "A-55" (proprietary diesel-water blend) would qualify as alternative fuels and would be exempt from state diesel fuel tax for a period of two years. After two years, the alternative fuels would be taxed only 6 cents/gallon (1/3 of the normal 18-cent fuel tax). Eliminating, then at least reducing the state excise fuel tax will provide a strong "positive driver" for the Biodiesel market. Meanwhile, the marine Biodiesel market in California remains the first (1993) and one of the few continuous commercial markets for Biodiesel in the United States. Over 7,000 gallons of Biodiesel have been sold to boaters in California by CytoCulture alone. Marine Biodiesel retails in California for $4.95 a gallon at fuel docks and marine supply stores where it is available. AEP sells their SoyGold product at several fuel docks in Southern California. CytoCulture has been distributing marine Biodiesel in Northern California since 1994 in 5-gallon containers (state approved, recyclable plastic containers). In 1997, CytoCulture set up 12 fuel docks and marine supply stores from Benicia to Monterey to promote the retail distribution of Biodiesel to boaters. CytoCulture is currently working to set up broader distribution of Biodiesel in returnable 5-gallon containers that would be purchased by the boater, then exchanged for a new container every time the customer bought another 5 gallons of the additive. As of April, 1999, Biodiesel can also be purchased and shipped from several of the producers listed on pages 26-27 in containers or by the 55-gallon drum. Marine Biodiesel is no longer retailed at fuel docks on the Chesapeake Bay, but CytoCulture and others are working to re-establish distribution through marine supply stores. Check the CytoCulture or NBB web sites for updates. Learning More About Marine Biodiesel The National Biodiesel Board can provide more general information on Biodiesel, including marine applications, by contacting them at: National Biodiesel Board P.O. Box 104898 Jefferson City, MO 65110-4898 Tel. 800-841-5849 Fax 573-635-7913
For a fun and extensive review of Biodiesel activities around the country and around the world, see Josh Tickell’s Veggie Van site at www.veggievan.org. From this site, you can order his book on making Biodiesel, get updated newsletters on Biodiesel and link up to other Biodiesel web sites around the world. CytoCulture may be contacted for information or questions regarding Marine Biodiesel programs in California, the results of our survey on biodiesel use in 100 recreational boats, Biodiesel specifications and testing protocols (European and American), California state laws that impact Biodiesel, and upcoming activities with the Marine Biodiesel League. CytoCulture is best reached via by telephone at 510-233-6660 or by email at: "Survey of 100 Recreational Boaters Using Biodiesel, 1994-1997" can be downloaded as a Word file along with this Handbook from the CytoCulture web site at: Questions, comments and suggestions regarding this Handbook are always welcome. This is the second edition (April 1999) of the original version drafted in September 1997 and printed April 1998. This report stems from over 5 years of experience testing and promoting the use of Biodiesel in recreational boats on the San Francisco Bay. The National Renewable Energy Laboratory (NREL, Golden, CO) sponsored the study and report under a subcontract (No. ACG-7-16688-01) supported by the U.S. Department of Energy. We would like to thank John Sheehan and Shaine Tyson (NREL) for their role in funding and overseeing the "Marine Biodiesel and Education Project for San Francisco Bay and Northern California". Special thanks are graciously extended to the following researchers and friends who patiently helped to edit and updated this revised edition of the Handbook: Ryan Werner, Esq. (Emeryville, CA and Biodiesel boater) Daryl Reese, M.S. (Utah State Univ., Pacific Biodiesel) Alan Weber (Weber Consulting and National Biodiesel Bd) Steve Howell (Marc IV Inc. and National Biodiesel Bd) Chris Sharp, Ph.D. (Southwest Research Institute, TX) Henning von Wedel, M.S. (Diesel engineer, Germany) Joshua Tickell (Veggie Van & Institute Renewable Energy) Bernard Wormgoor (Nauticus Surveyors) – Boater Hans Anderson (Corinthian Yacht Club Dock Master) Rita Gardner (Richmond Yacht Club) – Sailor/writer Breuer, C. (1994) Beitrag zur Verbrennung von Rapsol und Rapsmethylester in direkteinspritzenden Dieselmotoren (Report on Comparison of Rapeseed Oil and Rapeseed Methyl Ester in Direct Injection Diesel Motors), Doctoral Dissertation, Technical University, Hannover, Germany, 1994. CytoCulture (1997) Marine Biodiesel Survey Among 100 Recreational Boaters in San Francisco Bay Area and Northern California, Report for National Renewable Laboratory project sponsored by the US Department of Energy. CytoCulture (1997) Report on Regulatory and Tax Concerns Regarding Marine Biodiesel for Recreational Boats in California, Report for National Renewable Laboratory project sponsored by the US Dept. of Energy. CytoCulture (1997) Report on Establishing 12 Marine Biodiesel Distribution Points at Fuel Docks and Stores in No.California, Report for National Renewable Laboratory project sponsored by the US Dept. of Energy. Flechtner, M.K. and Gushee, D.E. (1993) Biodiesel Fuel: What Is It? Can it Compete? CRS Report for Congress, Congressional Research Service, The Library of Congress, Dec. 10, 1993. Howell, S. (1994) "Update Reports on Lubricity of Biodiesel Fuel, 1/26/94 and 3/10/94", Mark-IV Group, Kearny, MO, sponsored by the National SoyDiesel Development Board. Howell, S. (1997) Biodiesel Fuel Standard Making Progress, ASTM Standardization News, April, 1997, pp. 16-19. Knothe, G., Dunn, R.O., and Bagby, M.O. (1996) Technical Aspects of Biodiesel Standards, Inform 7 (8): 827-829. American Oil Chemists Society. Krawczyk, T. (1996) Biodiesel Alternative Fuel Makes Inroads But Hurdles Remain, Inform 7 (8): 801-815. American Oil Chemists Society. Leavitt, W. (1997) The Sweet Smell of Diesel, Trucking Technology, 1st Qtr 1997: 26-29. Merrill, P.N. (1994) Biodiesel Alert, 2 (5), published by the American Biofuels Association for the National Biodiesel Board, March 1994. Merrill, P.N. (1994) Biodiesel Alert, 2 (6), published by the American Biofuels Association for the National Biodiesel Board, April 1994. Perkins, L.A., Peterson, C.L. and Auld, D.L. (1994) Durability Testing of Transesterified Winter Rape Oil as Fuel in Small Bore, Multicylinder, DI, CI Engines, Report from the University of Idaho, National Center for Advanced Transportation Technology. Peterson, C.L. (1994), ed., Commercialization of Biodiesel: Establishment of Engine Warranties, Conference Proceedings November 9-10, 1994, National Center for Advanced Transportation Technology, University of Idaho, Moscow, ID. Peterson, C.L (1996) Development of Rapeseed Biodiesel for Use in High-Speed Diesel Engines, Progress Report for US Department of Energy, University of Idaho Department of Biological and Agricultural Engineering, May 31, 1996. Peterson, C.L. and Reece, D.L. (1996) Emissions Testing with Blends of Esters of Rapeseed Oil Fuel With and Without a Catalytic Converter, SAE Technical Paper Series, International Spring Fuels and Lubricants Meeting, Dearborne, MI, May 6-8, 1996. Peterson, C.L. and Reece, D.L. (1996) Emissions Characteristics of Ethyl and Methyl Ester Rapeseed Oil Compared with Low Sulfur Diesel Control Fuel in a Chassis Dynamometer Test of a Pickup Truck, Transactions of the ASAE, 39 (3): 805-816, May/June 1996. Peterson, C.L., Reece, D.L., Thompson, J.C., Beck, S.M., and Chase, C. (1996) Ethyl Ester of Rapeseed Used as a Biodiesel Fuel - A Case Study, Biomass and Bioenergy 10(5/6):331-336. Sharp, C.A. (1996) Emissions and Lubricity Evaluation of Rapeseed Derived Biodiesel Fuels, Southwest Research Institute Study (San Antonio, TX) sponsored by the Department of Energy and University of Idaho; presented at DOE Biodiesel Emissions Testing Meeting, Seattle, WA, November 25, 1996. Shay, E.G. (1993) Diesel Fuel from Vegetable Oils: Status and Opportunities. Biomass and Bioenergy, 4(4): 227-242. [Report from the National Academy of Sciences] Sheehan, J. and Duffield, J. (1996, 1998) "Life Cycle Assessment of Petroleum-Base Diesel Fuel and Biodiesel" report issued by the Biotechnology Center of the National Renewable Energy Laboratory under contract with the US Department of Energy, Golden Colorado, July 1996; June, 1998. Teal, R. (1995) Introducing Biodiesel into the Marine Market Phase 1: Florida Keys, Report for National Biodiesel Board by Global Cartographics, Inc., Marathon, FL, October 16, 1995. Tyson, K. Shain (1998) Biodiesel Research Progress, National Renewable Energy Laboratory, Golden, CO. Varese, R. and Varese, M. (1996) Methyl Ester Biodiesel: Opportunity or Necessity? Inform 7 (8): 816-824. American Oil Chemists Society. Womac, A.R., Crouch, J.A., Easterly, C., and Strange, R.J. (1996) Evaluation and Optimization of a 110-HP Volvo Marine Diesel Engine Using Soydiesel, Report prepared for the National Biodiesel Board by the Department of Agricultural Engineering, University of Tennessee, Knoxville, TN, August 13, 1996. Womac, A.R., Strange, R.J., Crouch, J.A., and Easterly, C. (1996) Power, Emissions, and Bioresponse of Biodiesel in a Marine Environment, Report prepared for the National Biodiesel Board by the Department of Agricultural Engineering, University of Tennessee, Knoxville, TN. Zhang, X., Peterson, CL, Reece, D., Moller, G. and Haws, R. (1995) University of Idaho Technical Report on "Biodegradability of Biodiesel in the Aquatic Environment". |
||
 |
||
